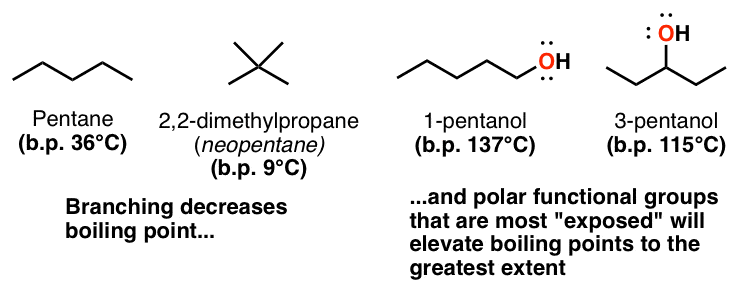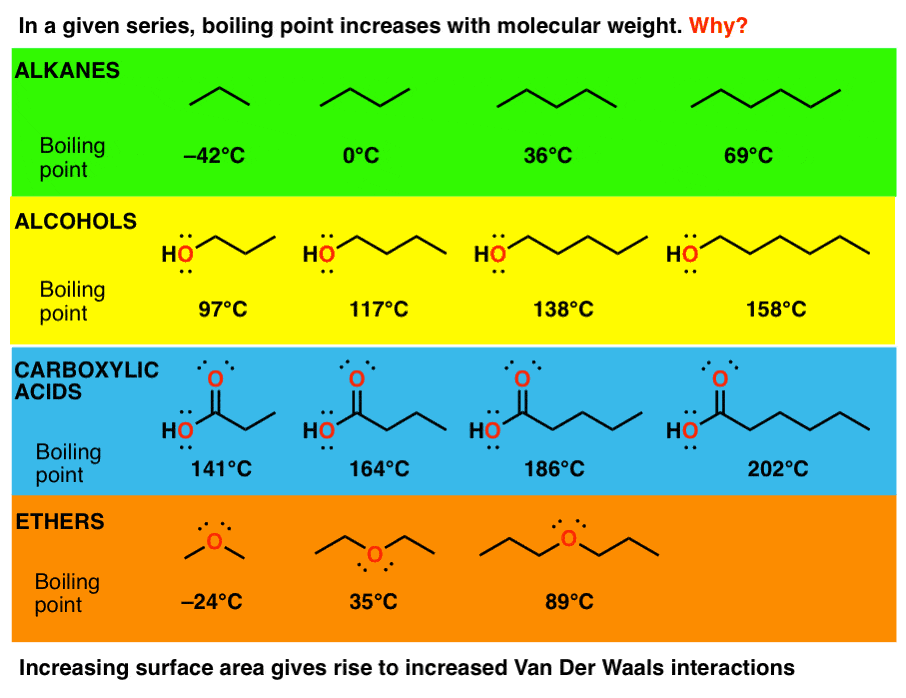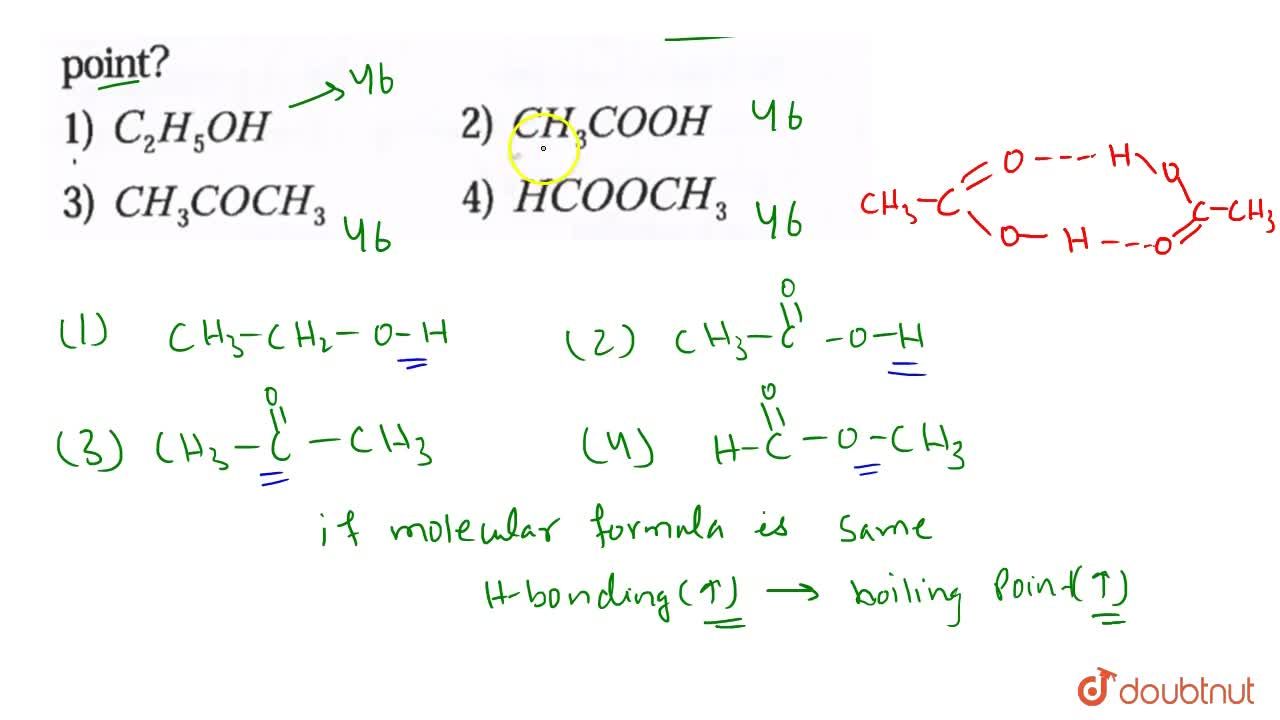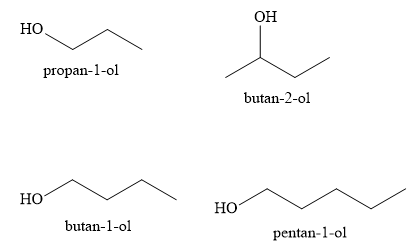
Arrange the following compounds in increasing order of boiling points . Propan-1-ol , butan-I-ol , butan-2-ol, pentan-1-ol. A) Propan-l-ol, butan-2-ol,butan-1-ol ,pentan-1-ol B) Propan-l-ol, butan-1-ol,butan-2-ol ,pentan-1-ol C) Pentan-1-ol , butan-2 ...

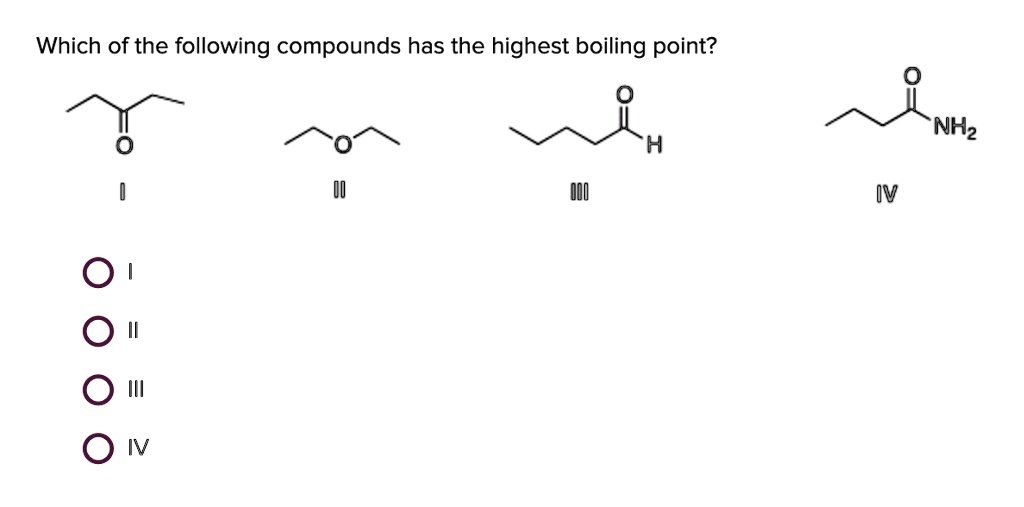
Rank these compounds by boiling point. 2. Select the compound that has the highest boiling point, based on that compound's dominant intermolecular forces. a. Pentanal b. 1-fluoropentane c. Diethyl eth | Homework.Study.com
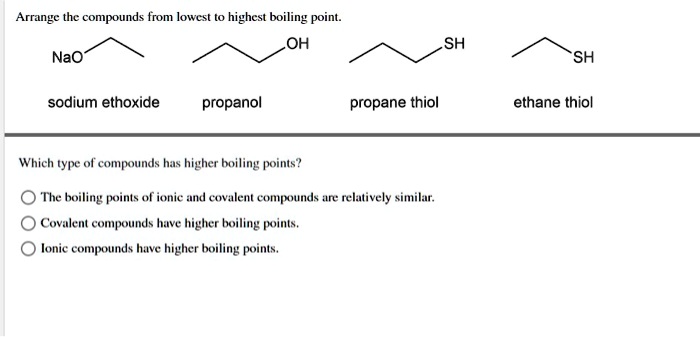
SOLVED: Arrange the compounds from lowest to highest boiling poin OH NaO SH SH sodium ethoxide propanol propane thiol ethane thiol Which type ol compounds has higher boiling' points? The boiling pints

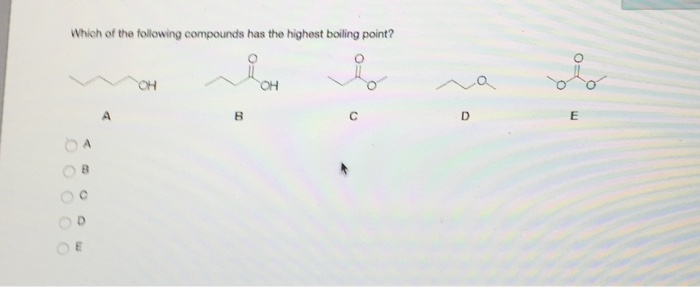
OneClass: . Which one of the following compounds will have the highest boiling poing? A) CH CH CH CH ...

Which group of elements has the highest boiling point? * IIIa (13) IIa (2) Ia (1) IVa (14) Transition - Brainly.com
![Which among the following has the highest boiling point?A. \\[{{C}}{{{H}}_3}{{C}}{{{H}}_2}{{C}}{{{H}}_2}{{C}}{{{H}}_2}{{Cl}}\\]B. ${\\left( {{{C}}{{{H}}_3}} \\right)_2}{{CHC}}{{{H}}_2}{{Cl}}$C. ${\\left( {{{C}}{{{H}}_3}} \\right)_3}{{C}} - {{Cl}}$D ... Which among the following has the highest boiling point?A. \\[{{C}}{{{H}}_3}{{C}}{{{H}}_2}{{C}}{{{H}}_2}{{C}}{{{H}}_2}{{Cl}}\\]B. ${\\left( {{{C}}{{{H}}_3}} \\right)_2}{{CHC}}{{{H}}_2}{{Cl}}$C. ${\\left( {{{C}}{{{H}}_3}} \\right)_3}{{C}} - {{Cl}}$D ...](https://www.vedantu.com/question-sets/27b4dbf3-0411-41ad-b019-f7310d8431d53412833081885436125.png)