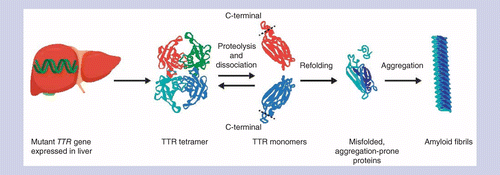
A phase II, open-label, extension study of long-term patisiran treatment in patients with hereditary transthyretin-mediated (hATTR) amyloidosis

Prediction of the complex structure of patisiran (Onpattro). The input... | Download Scientific Diagram

Leading RNA Interference Therapeutics Part 1: Silencing Hereditary Transthyretin Amyloidosis, with a Focus on Patisiran | SpringerLink

The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs | Nature Nanotechnology

Difference in the lipid nanoparticle technology employed in three approved siRNA (Patisiran) and mRNA (COVID-19 vaccine) drugs - ScienceDirect
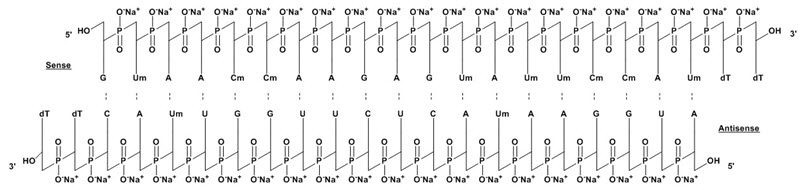
These highlights do not include all the information needed to use ONPATTRO® safely and effectively. See full prescribing information for ONPATTRO. ONPATTRO (patisiran) lipid complex injection, for intravenous useInitial U.S. Approval: 2018

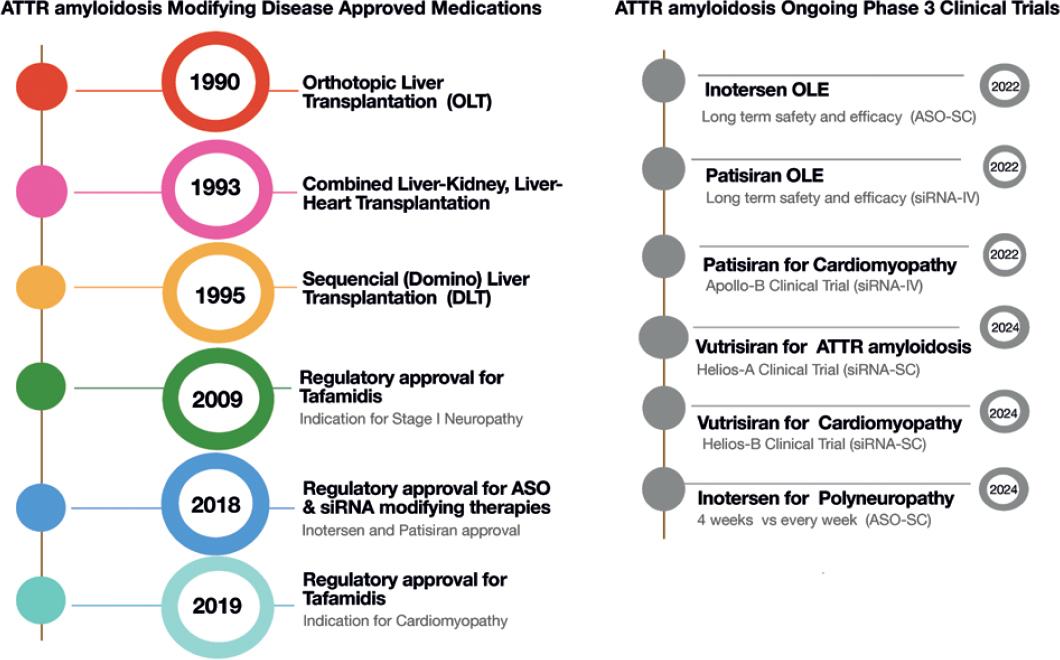
Alnylam Announces First-Ever FDA Approval of an RNAi Therapeutic, ONPATTRO™ (patisiran) for the Treatment of the Polyneuropathy of Hereditary Transthyretin-Mediated Amyloidosis in Adults | Business Wire
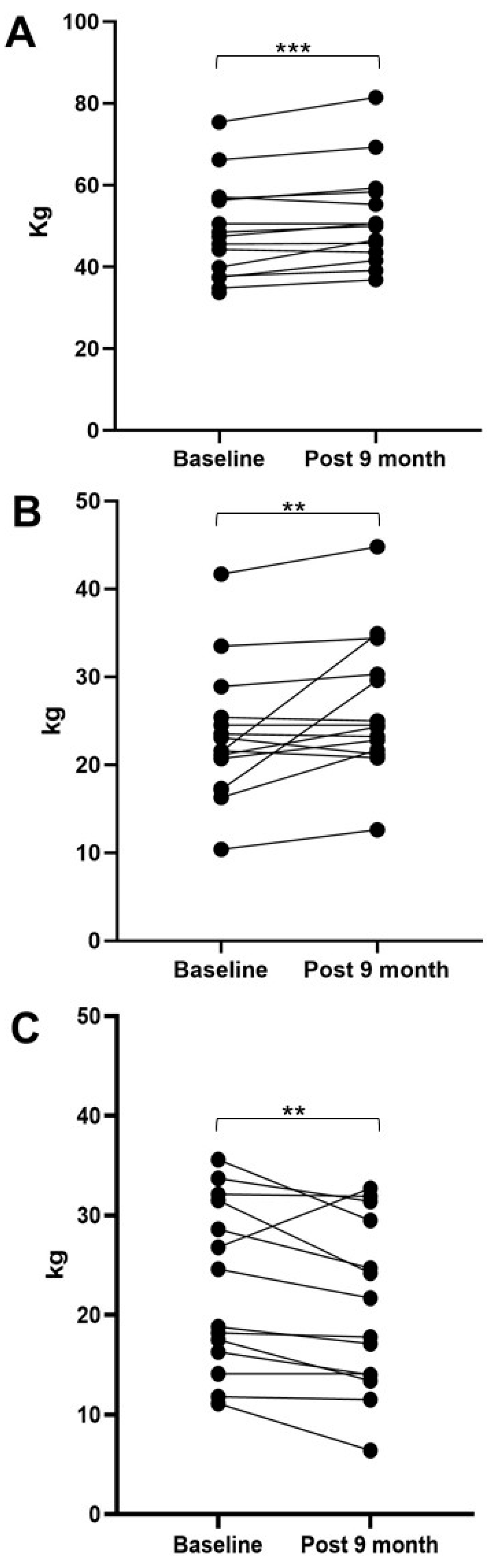
Biomedicines | Free Full-Text | Patisiran Enhances Muscle Mass after Nine Months of Treatment in ATTRv Amyloidosis: A Study with Bioelectrical Impedance Analysis and Handgrip Strength

Patisiran treatment in patients with hereditary transthyretin-mediated amyloidosis with polyneuropathy after liver transplantation - American Journal of Transplantation