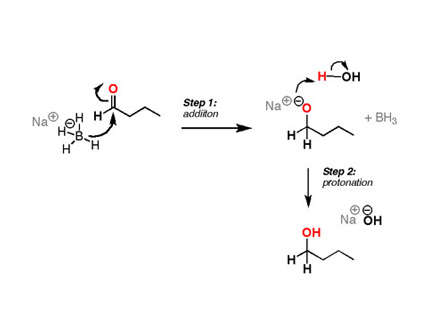
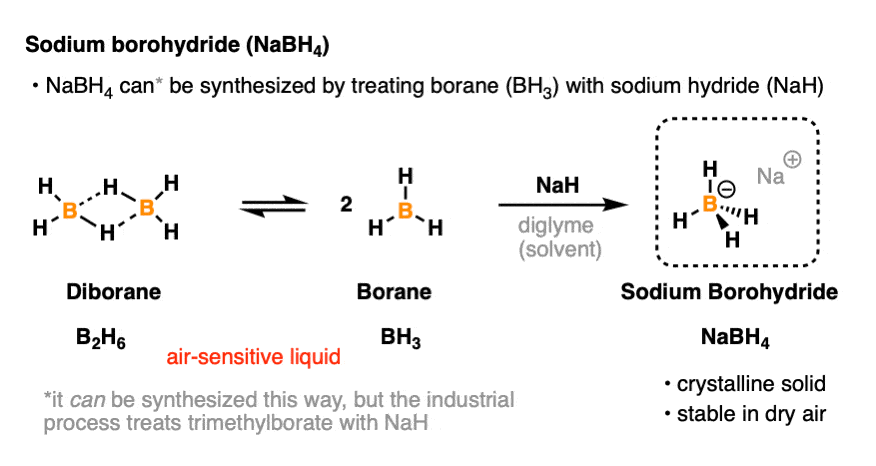
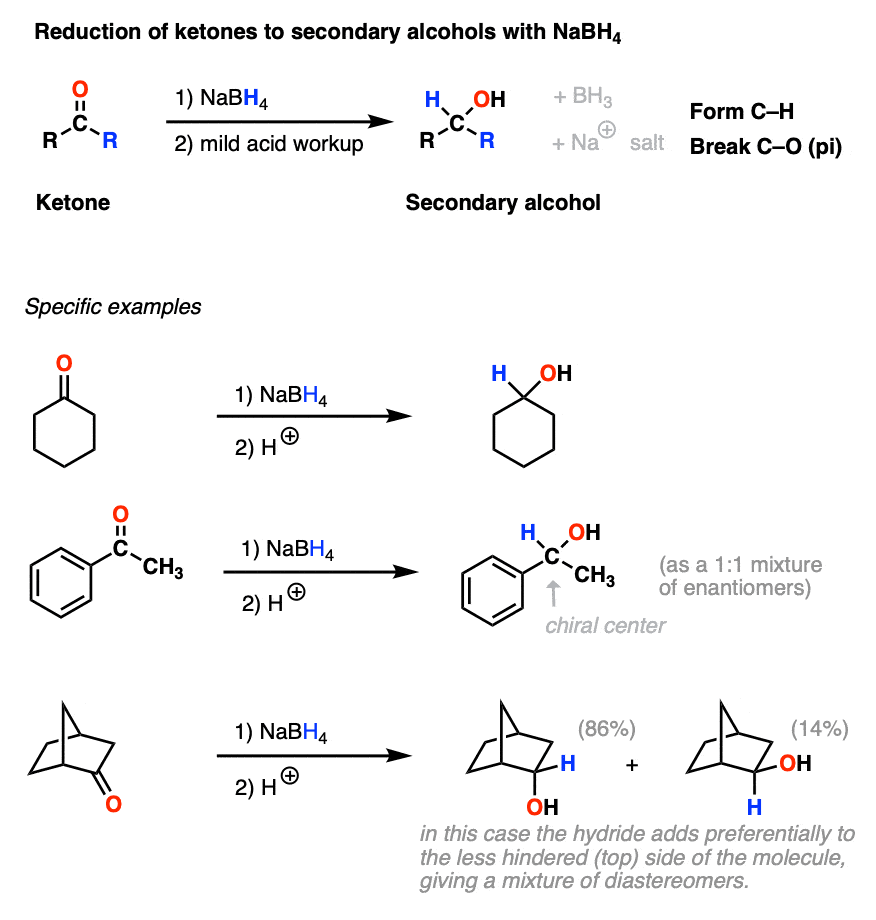
Stabilization of NaBH4 in Methanol Using a Catalytic Amount of NaOMe. Reduction of Esters and Lactones at Room Temperature without Solvent-Induced Loss of Hydride | The Journal of Organic Chemistry
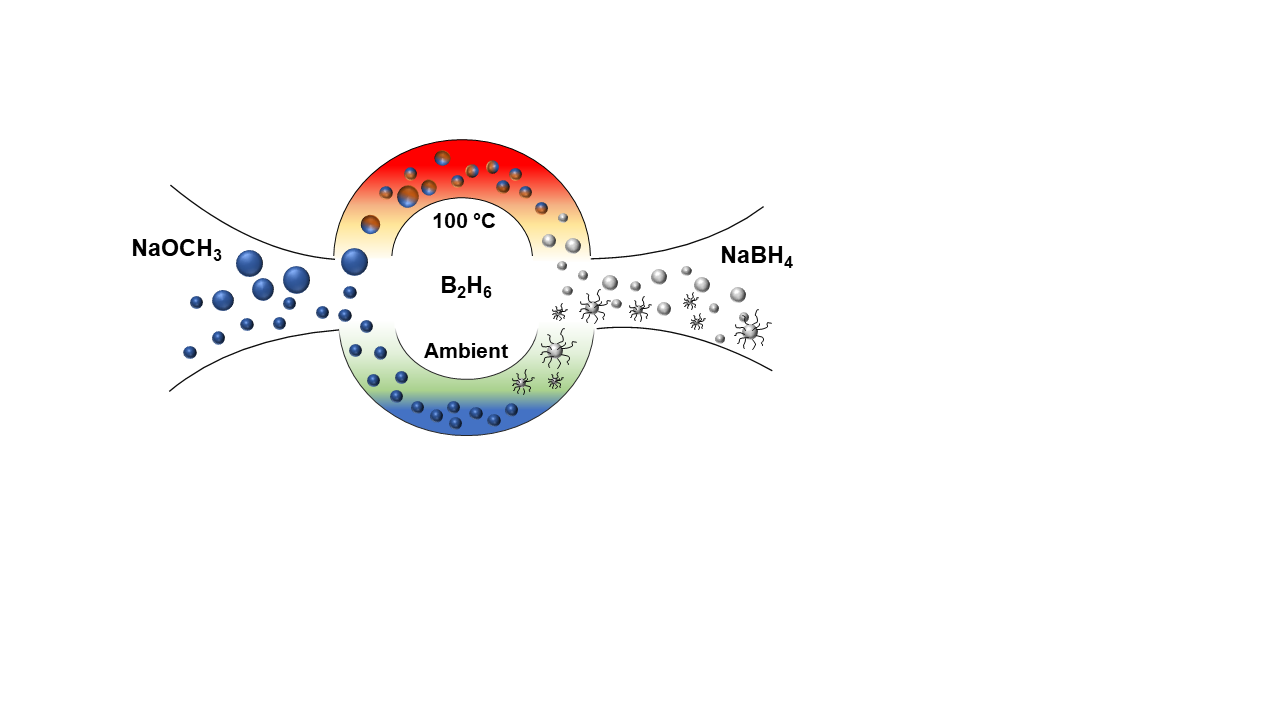
Energies | Free Full-Text | Direct Synthesis of NaBH4 Nanoparticles from NaOCH3 for Hydrogen Storage

Improved Procedures for the Generation of Diborane from Sodium Borohydride and Boron Trifluoride | Inorganic Chemistry
Energies | Free Full-Text | Direct Synthesis of NaBH4 Nanoparticles from NaOCH3 for Hydrogen Storage

A Co2B Mediated NaBH4 Reduction Protocol Applicable to a Selection of Functional Groups in Organic Synthesis - Lundevall - 2018 - European Journal of Organic Chemistry - Wiley Online Library

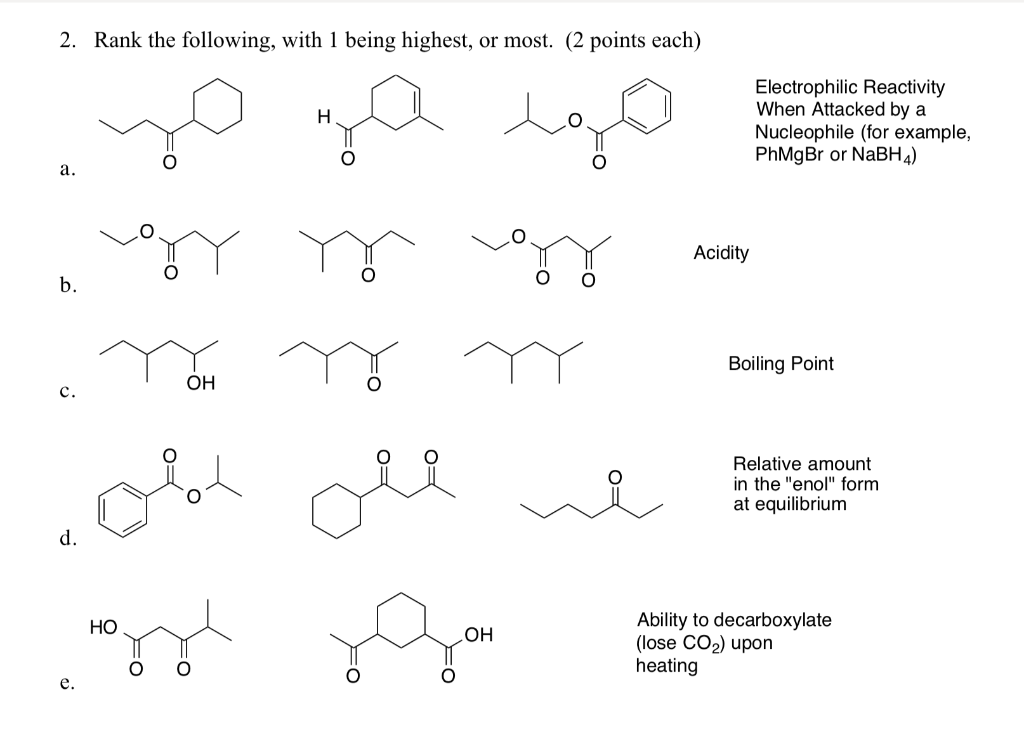
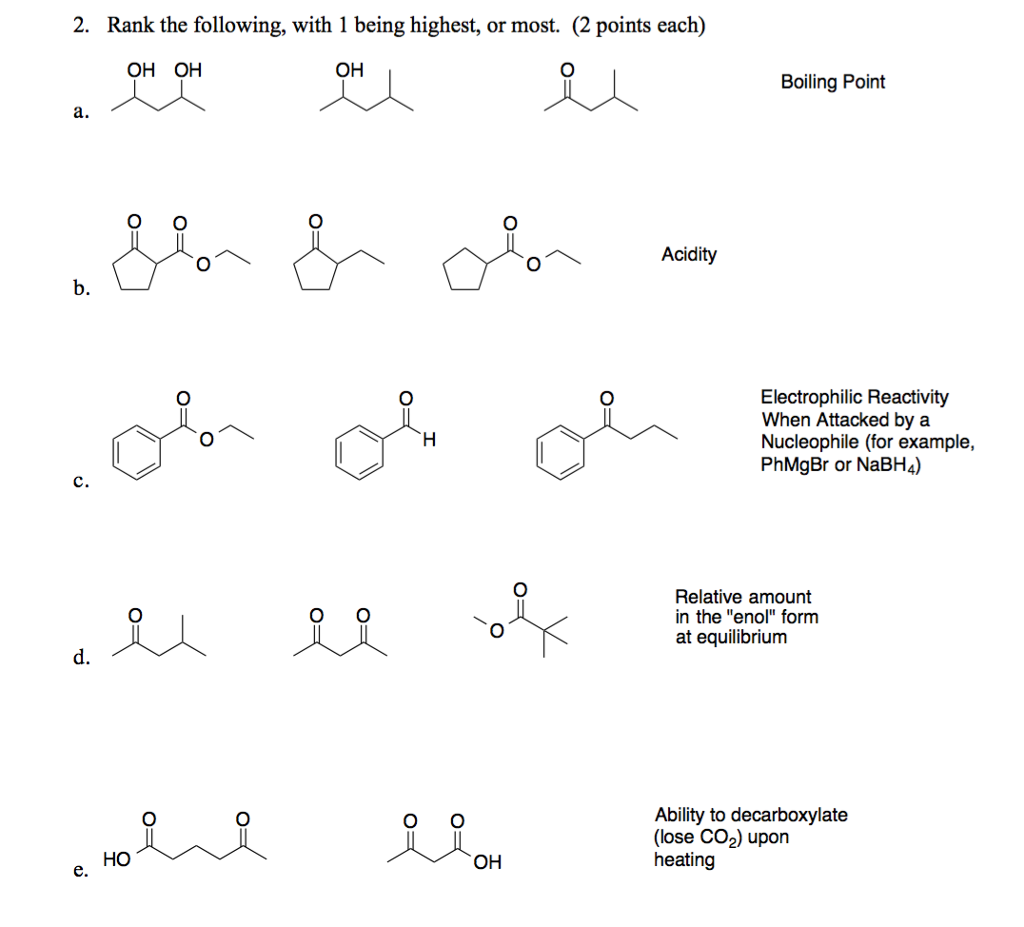
SOLVED: a) Compound A, is reduced by sodium borohydride to give products B and € as shown in the reaction below: OH OHI NaBH EtOH A B Give the IUPAC name (including