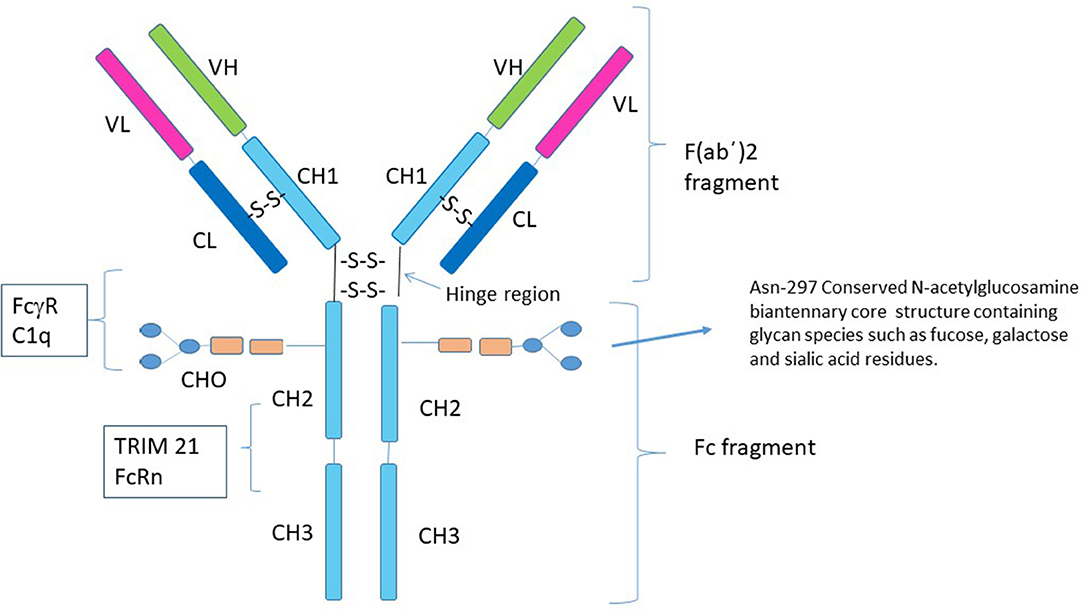
Structure of the streptococcal endopeptidase IdeS, a cysteine proteinase with strict specificity for IgG | PNAS
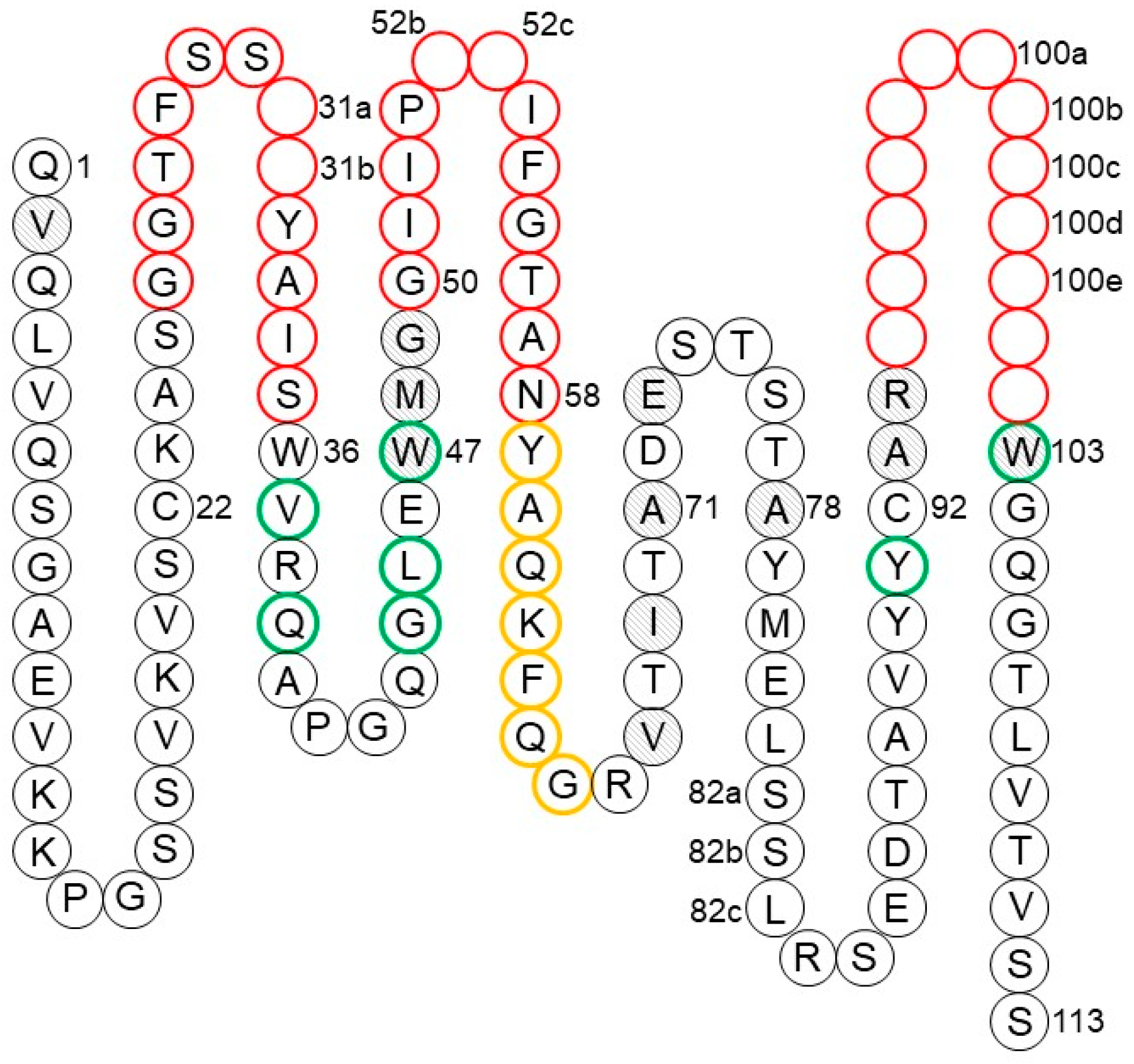
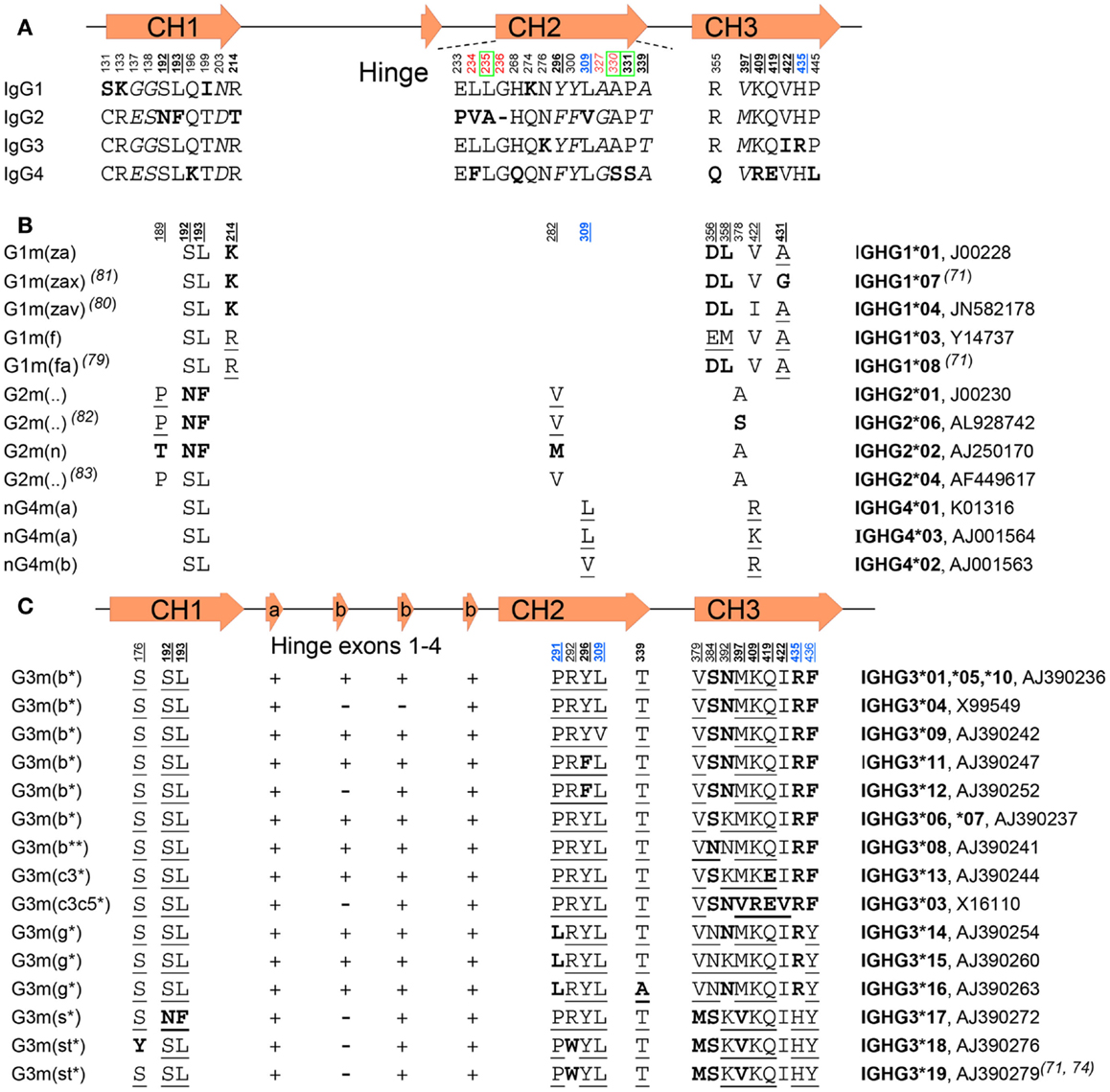
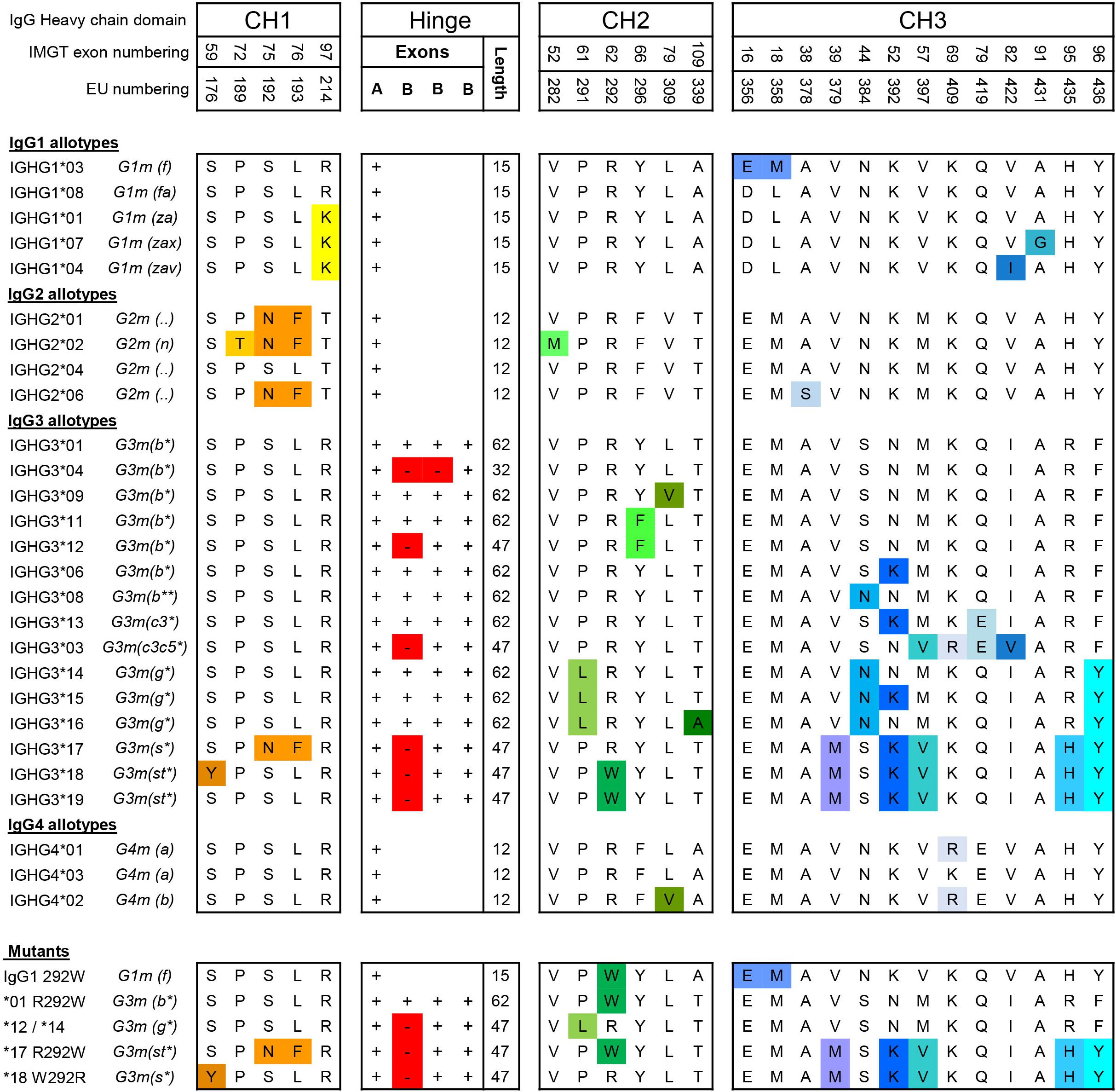
![PDF] The Origins, Specificity, and Potential Biological Relevance of Human Anti-IgG Hinge Autoantibodies | Semantic Scholar PDF] The Origins, Specificity, and Potential Biological Relevance of Human Anti-IgG Hinge Autoantibodies | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/9e5f002374ecf7abd15e4aeacda63aa75162f32a/6-Figure1-1.png)
PDF] The Origins, Specificity, and Potential Biological Relevance of Human Anti-IgG Hinge Autoantibodies | Semantic Scholar

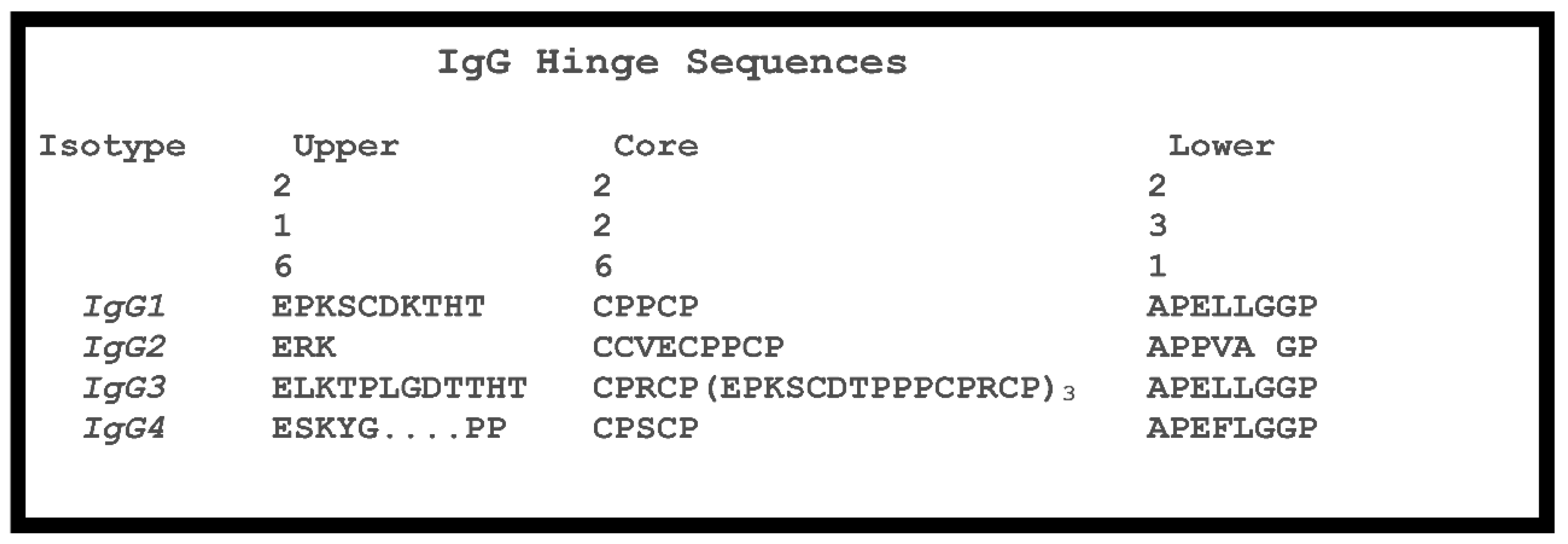
A , Comparison of the amino acid sequence for CH1 and hinge regions for... | Download Scientific Diagram
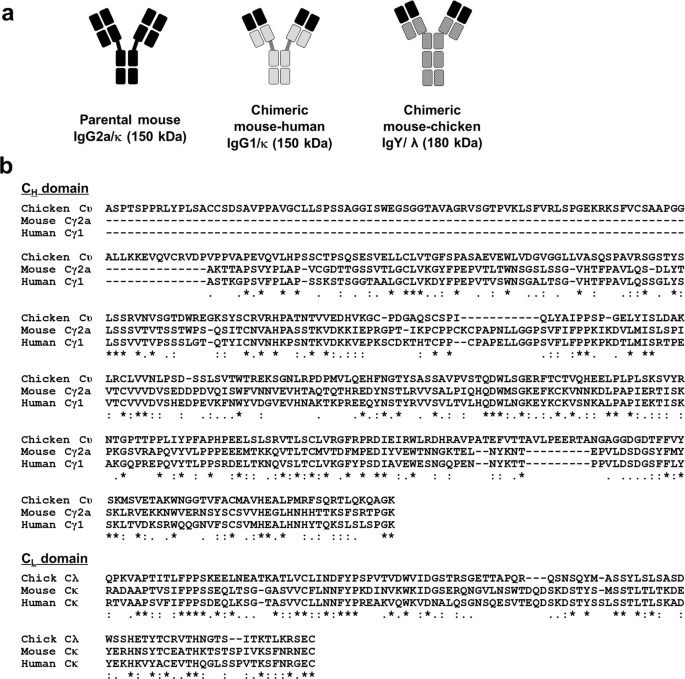
Antigen-binding affinity and thermostability of chimeric mouse-chicken IgY and mouse-human IgG antibodies with identical variable domains | Scientific Reports

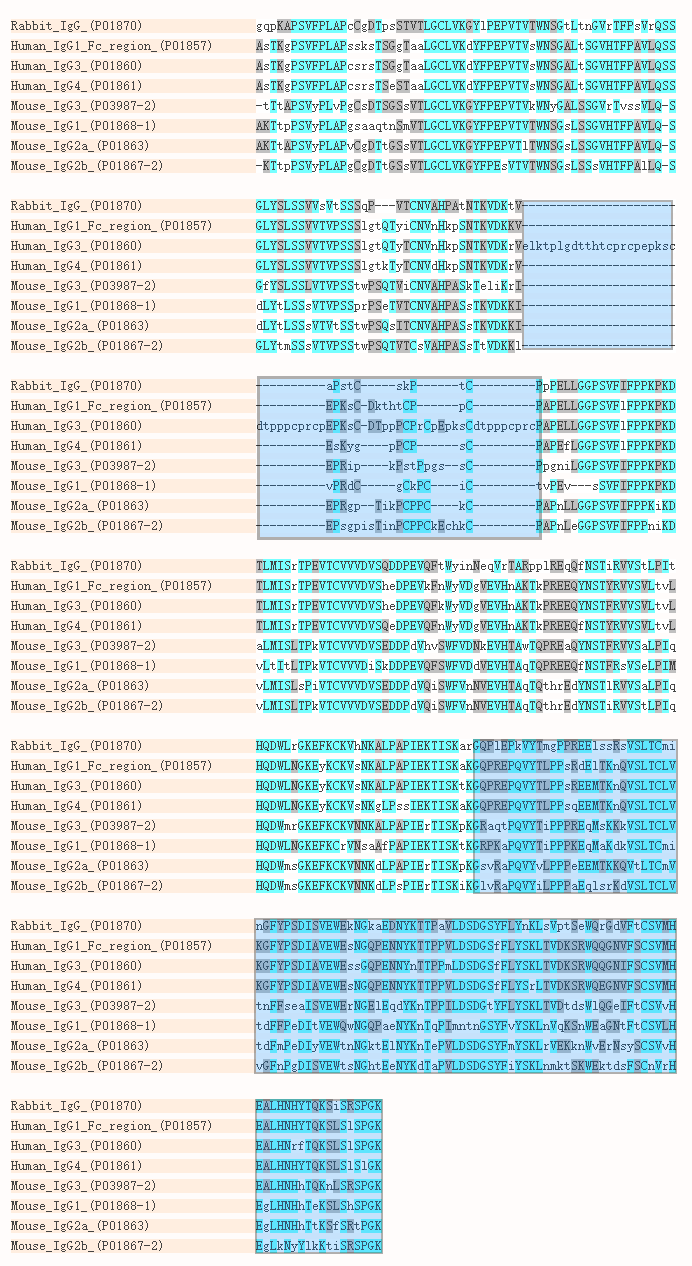
Amino acid sequence alignment of the Fc region of eqIgGs. The sequence... | Download Scientific Diagram

Structural basis for binding of human IgG1 to its high-affinity human receptor FcγRI | Nature Communications

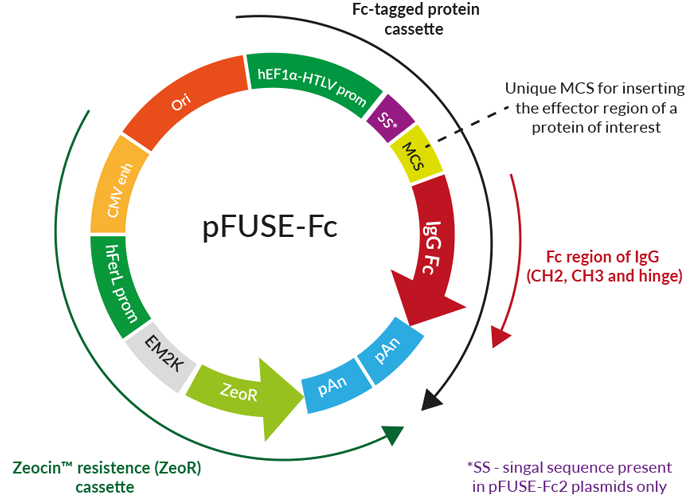
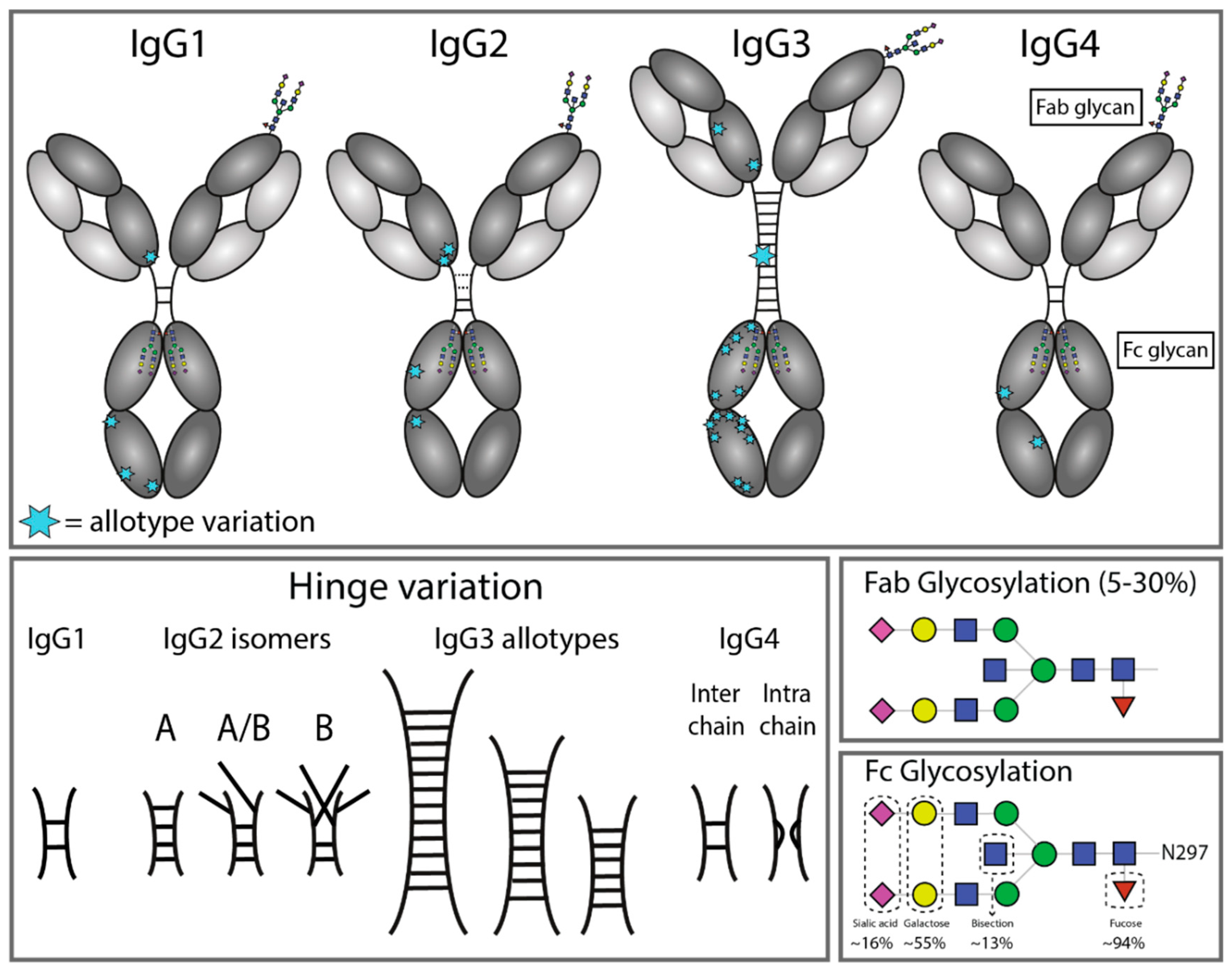
Engineered IgG1‐Fc – one fragment to bind them all - Lobner - 2016 - Immunological Reviews - Wiley Online Library

Human IgG1, IgG3, and IgG3 Hinge-Truncated Mutants Show Different Protection Capabilities against Meningococci Depending on the Target Antigen and Epitope Specificity | Clinical and Vaccine Immunology
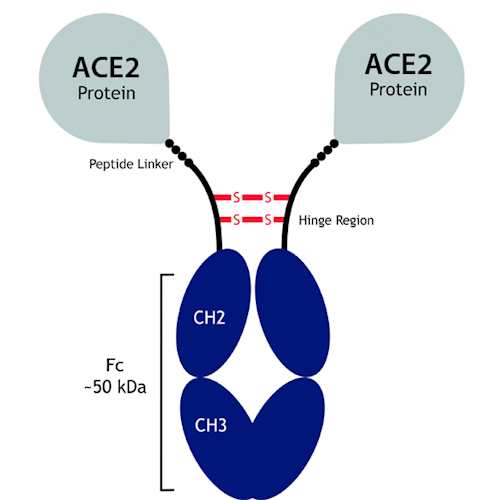
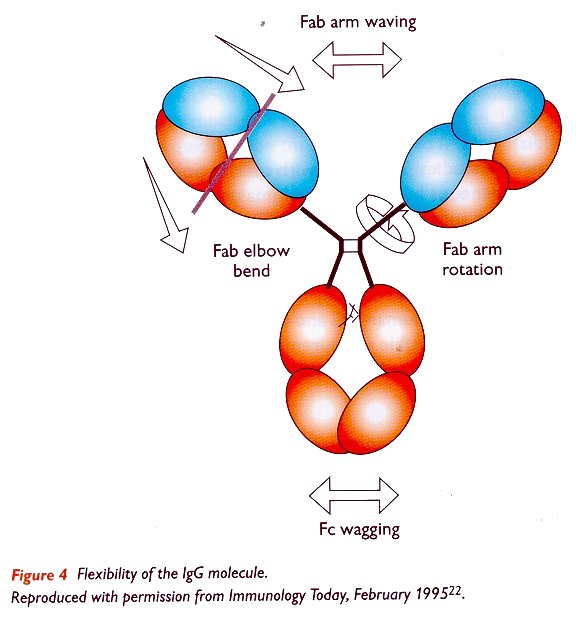
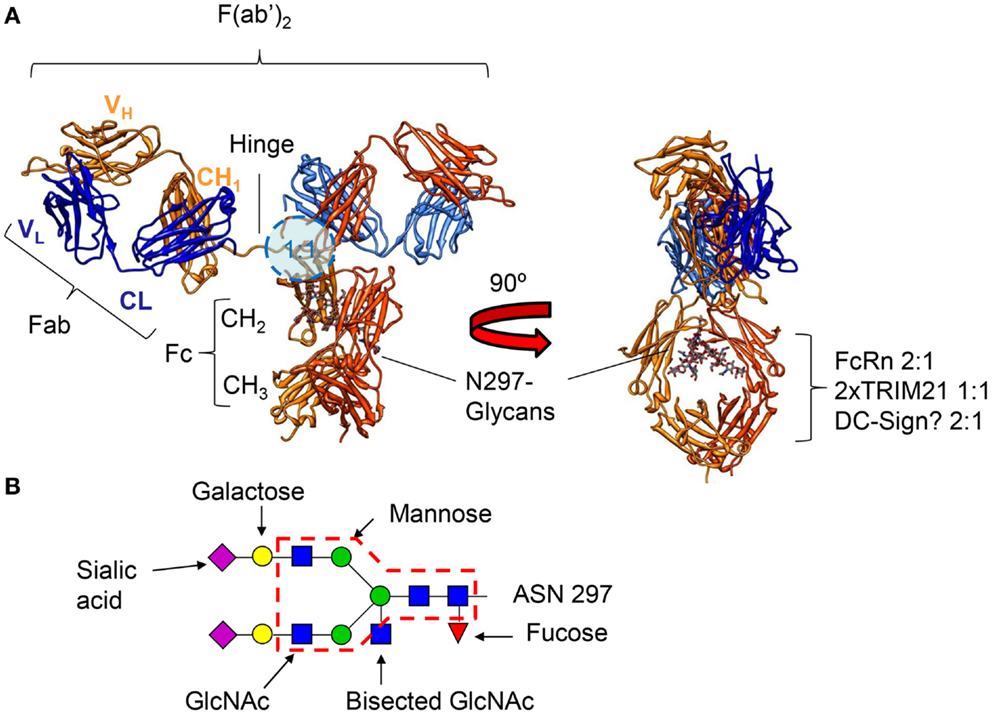
Antibodies | Free Full-Text | Antibody Structure and Function: The Basis for Engineering Therapeutics

Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports

Cross-reactivity of mouse IgG subclasses to human Fc gamma receptors: Antibody deglycosylation only eliminates IgG2b binding - ScienceDirect
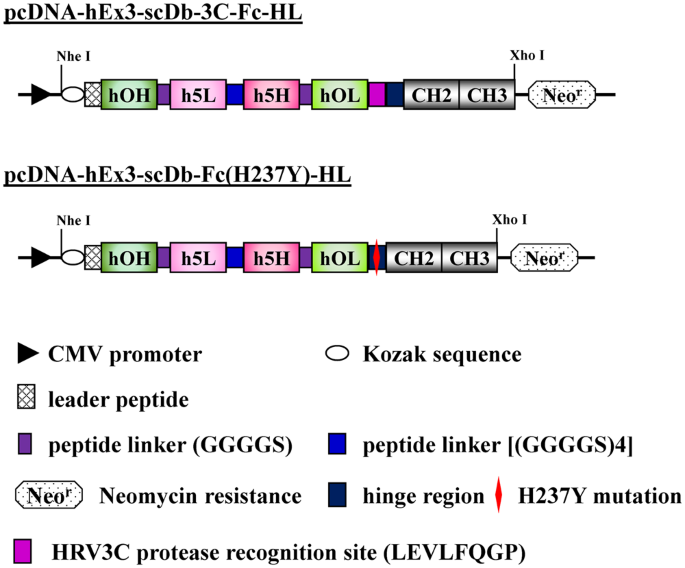
Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports