Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology

Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology

A) Three tags, i.e. 10x-His, FLAG and HA, and one proteolytic site for... | Download Scientific Diagram
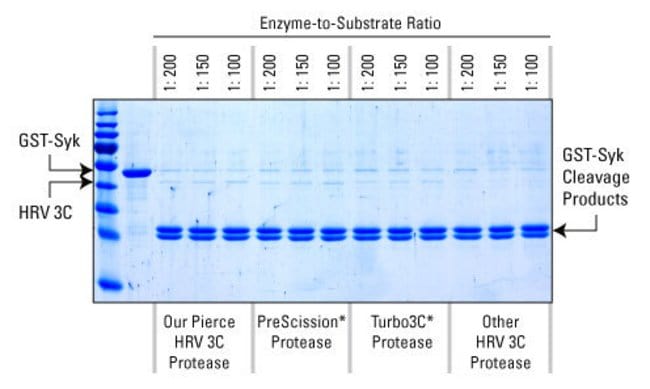
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE

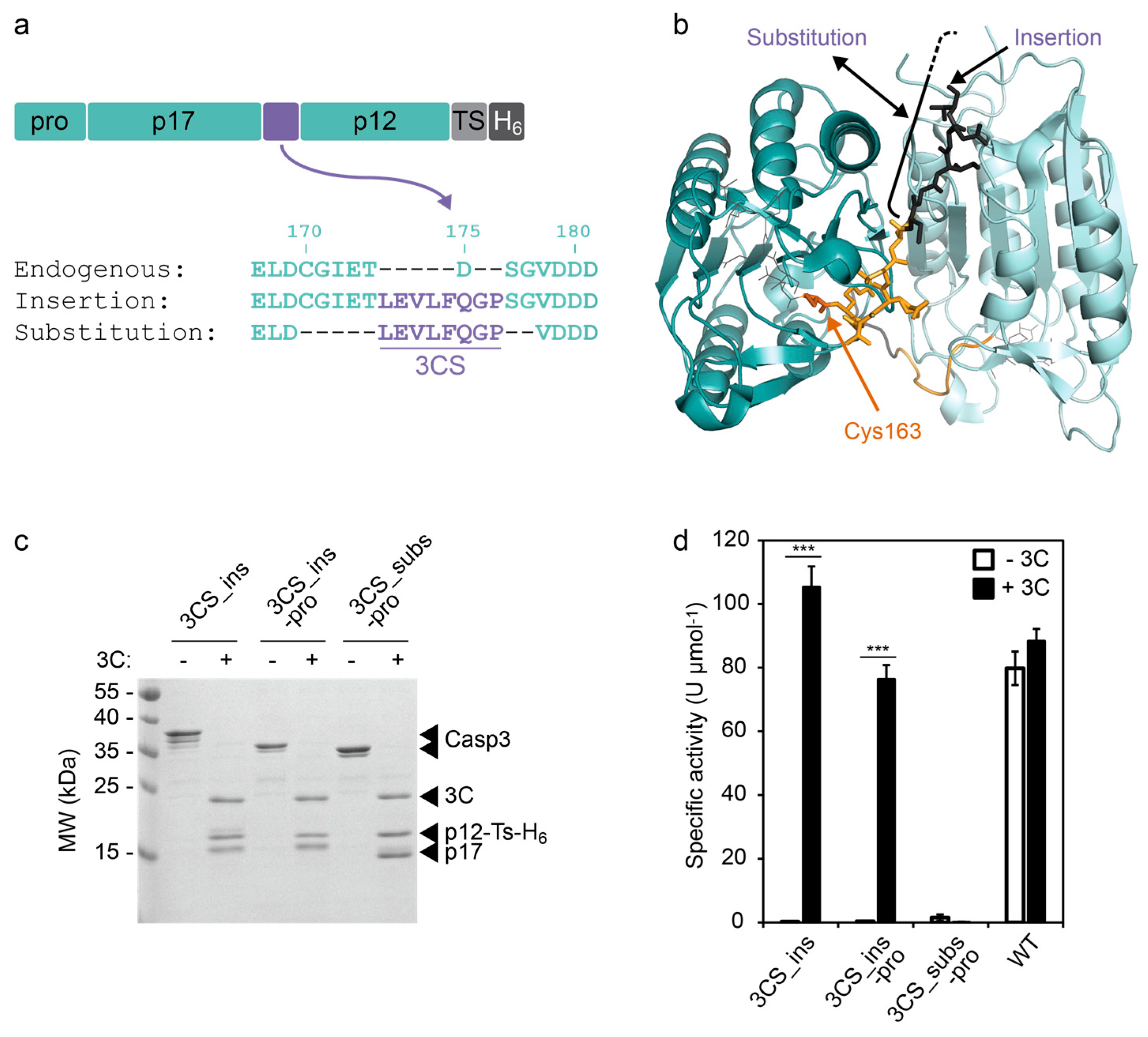
Directed evolution of the 3C protease from coxsackievirus using a novel fluorescence-assisted intracellular method