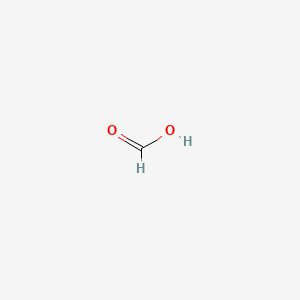
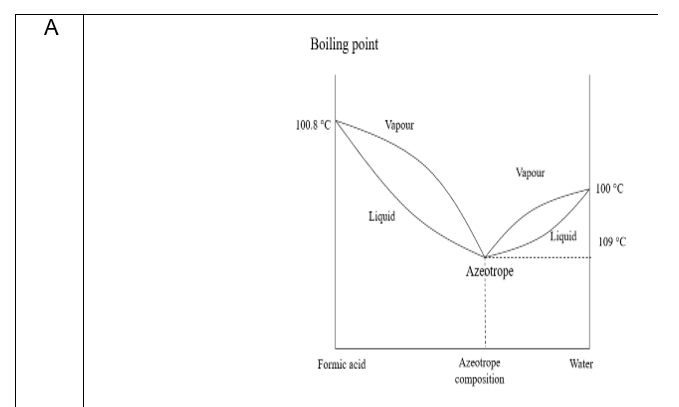
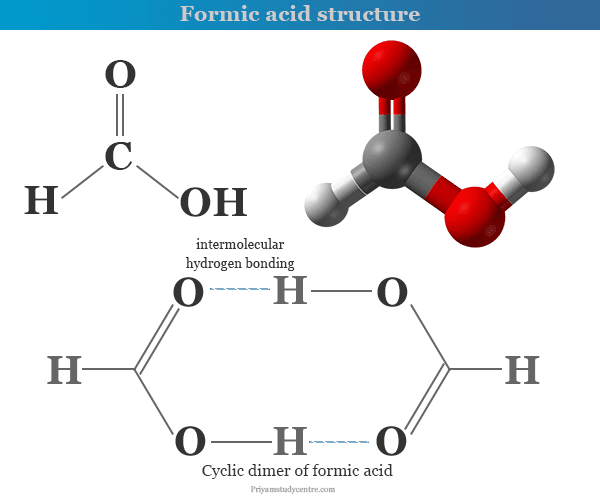
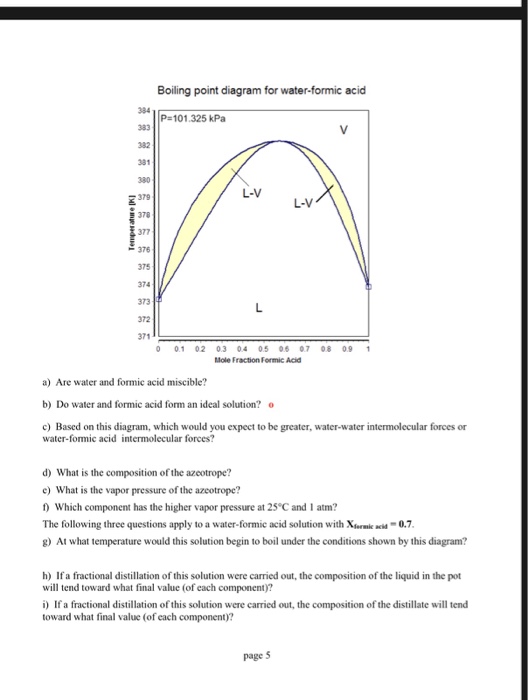
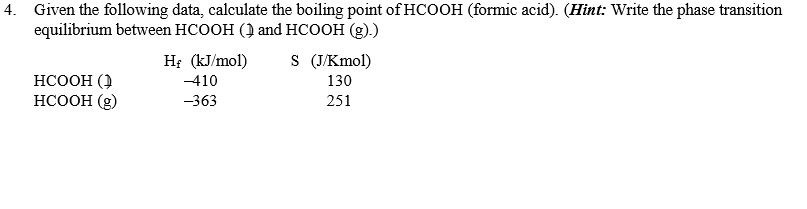
The freezing point depression of a 0.109 M aq. solution of formic acid is - 0.21^∘C . The equilibrium constant for the reaction is 1.44 × 10^-x . HCOOH (aq) H^+(aq) +

Formic Acid to Power towards Low‐Carbon Economy - Dutta - 2022 - Advanced Energy Materials - Wiley Online Library