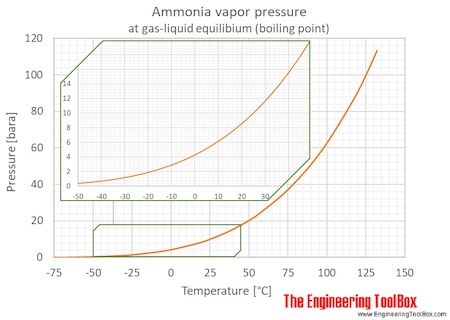
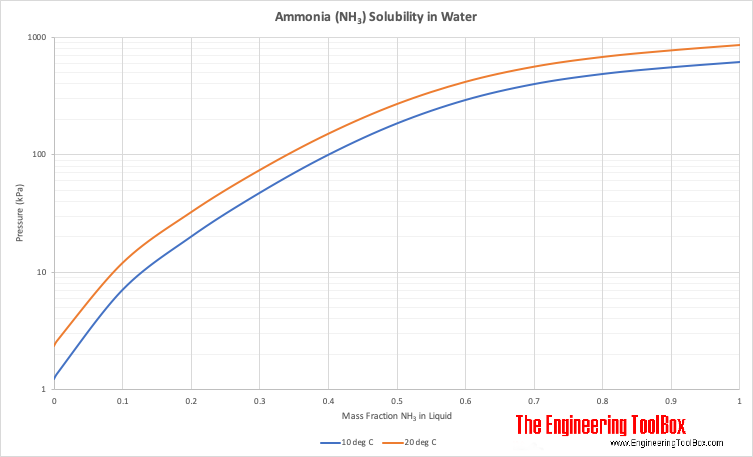
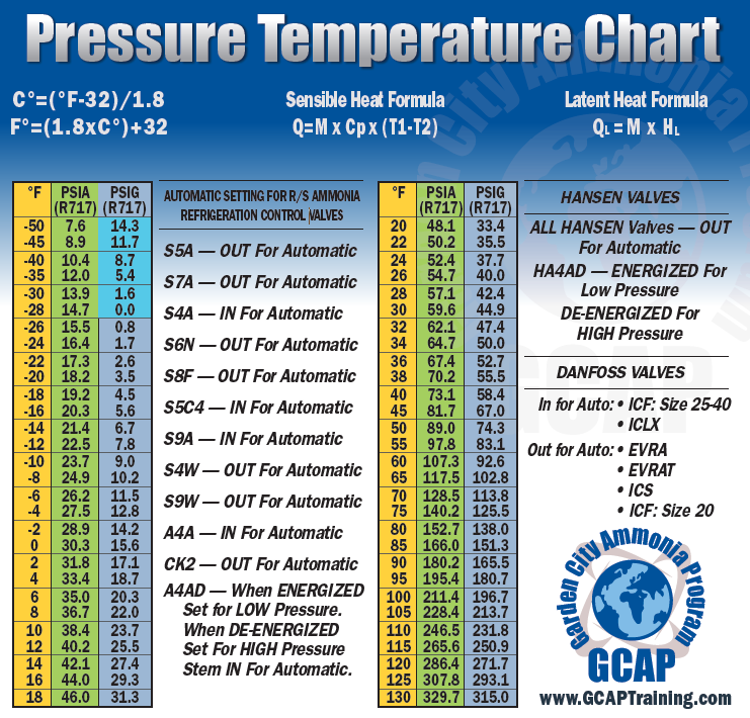
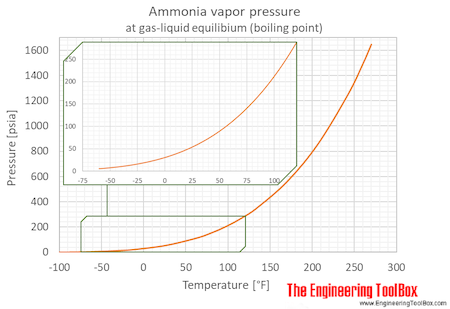
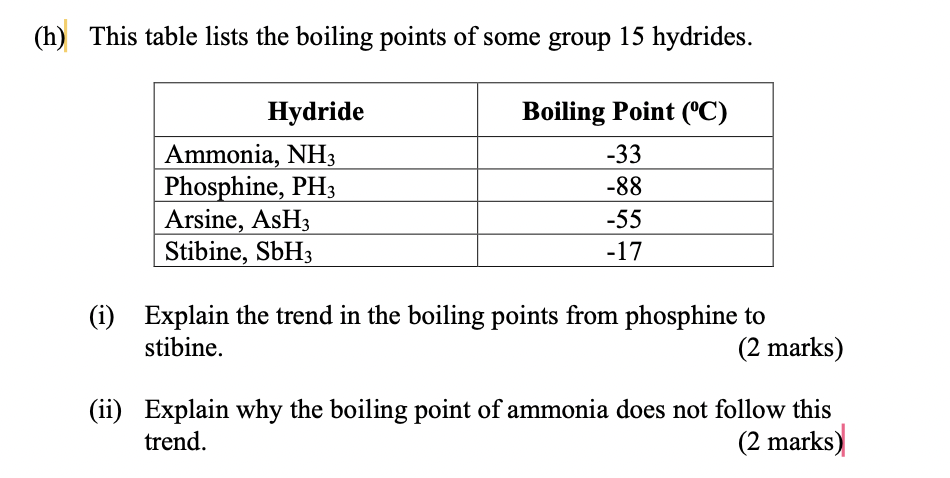
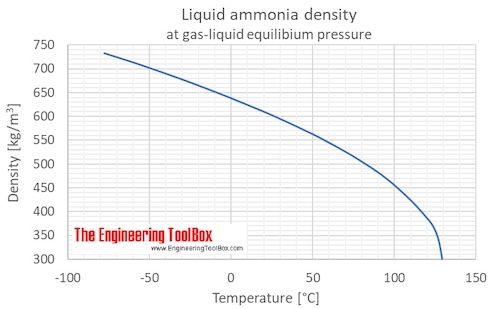
Ocean thermal energy conversion uses ammonia with a boiling point around -33 f, but the cold seawater in the condenser is well above that temperature. How is the ammonia condensed? - Quora
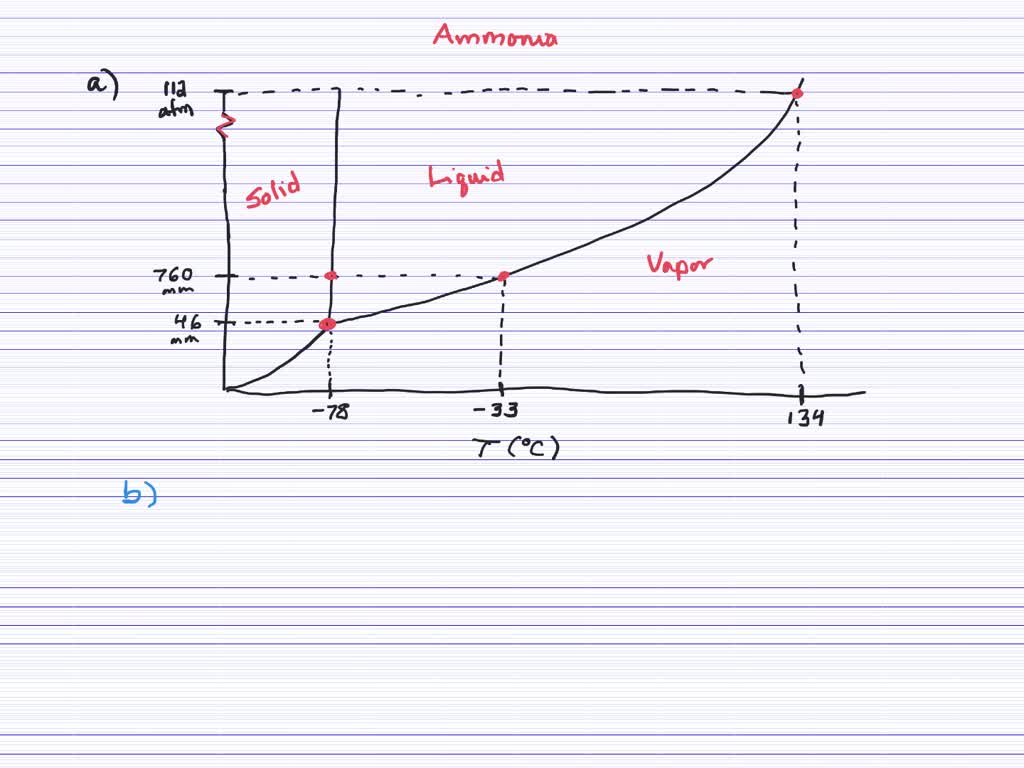
SOLVED:Given the following data about ammonia, normal boiling point =-33^∘C normal melting point =-78^∘C triple point =-78^∘C at 46 mmHg critical point =132^∘C at 112 atm (a) Construct an approximate phase diagram
Anhydrous Ammonia (Nh3) Boiling Point: -33.34 Degree C at Best Price in Mumbai | Jaysons Chemical Industries

Energies | Free Full-Text | Experimental Investigation of High-Pressure Liquid Ammonia Injection under Non-Flash Boiling and Flash Boiling Conditions

Ammonia has much higher boiling point than phosphine due to _____ . | 12 | P-BLOCK GROUP 15 ELEM... - YouTube