Directed evolution of the 3C protease from coxsackievirus using a novel fluorescence-assisted intracellular method
Frontiers | Identification of Cleavage Sites Recognized by the 3C-Like Cysteine Protease within the Two Polyproteins of Strawberry Mottle Virus

Uncovering targets of the Leader protease: Linking RNA‐mediated pathways and antiviral defense - Saiz - 2021 - WIREs RNA - Wiley Online Library
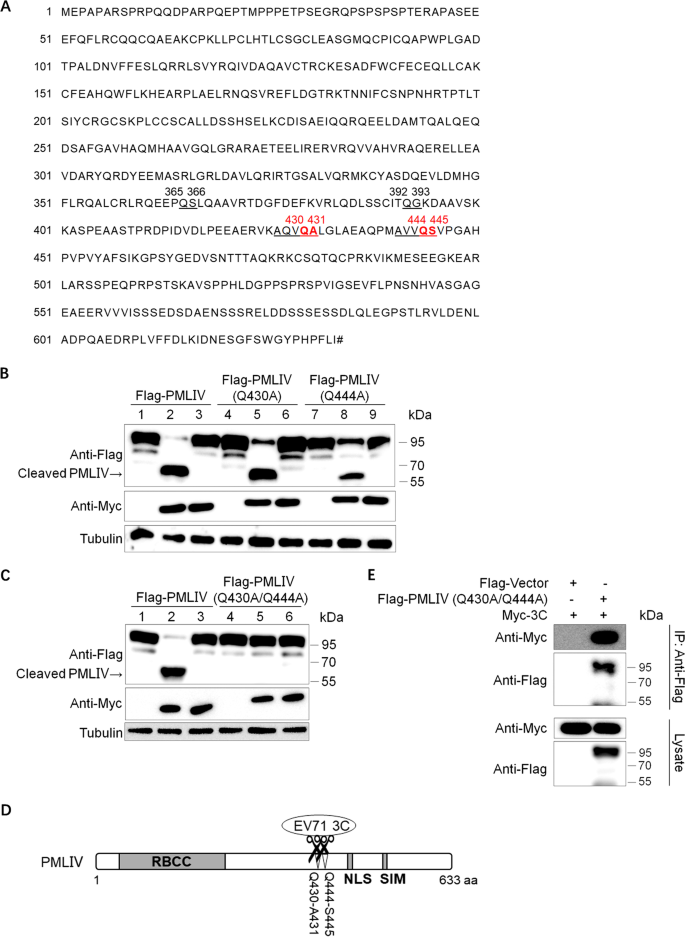
3C protease of enterovirus 71 cleaves promyelocytic leukemia protein and impairs PML-NBs production | Virology Journal | Full Text

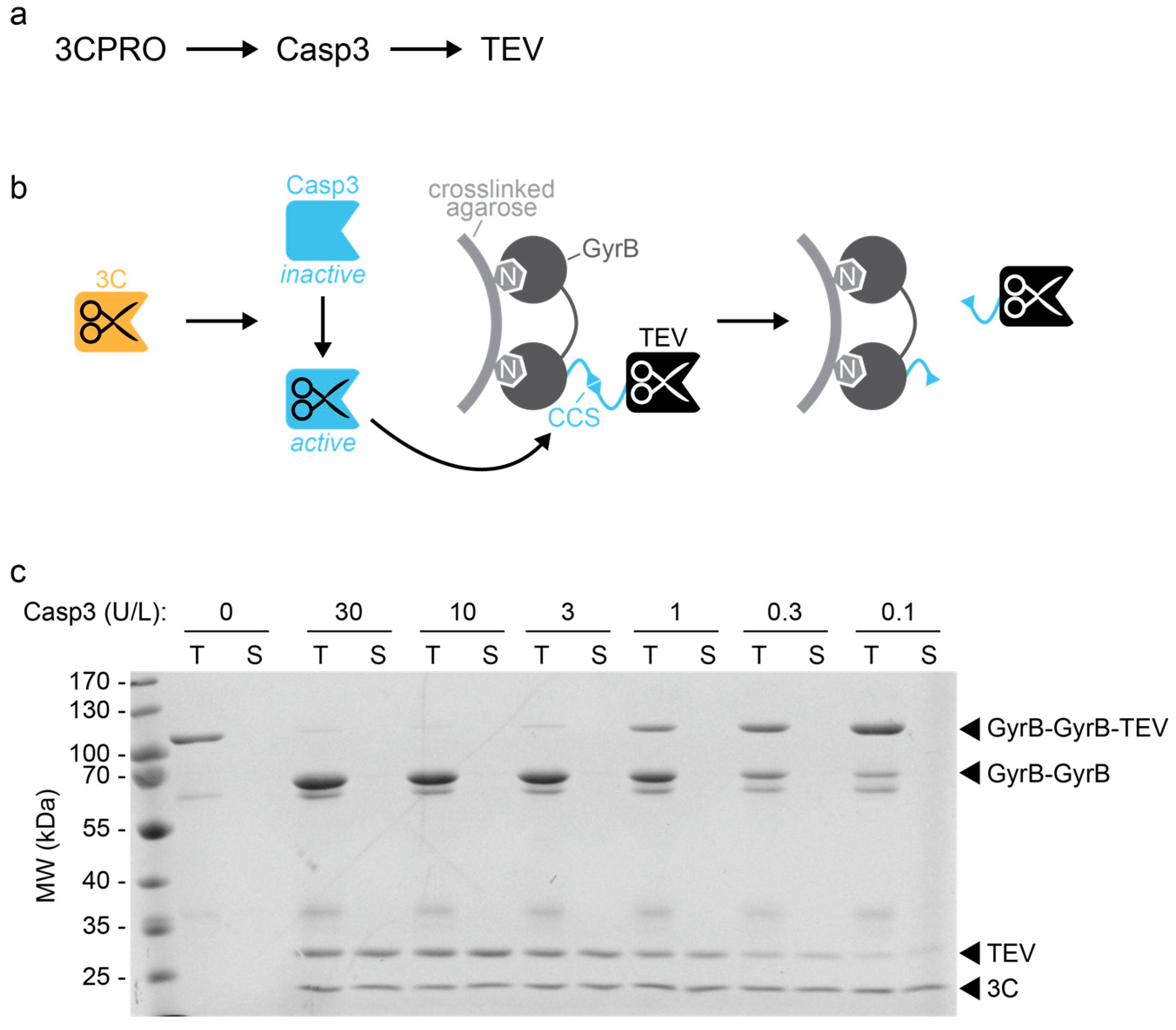
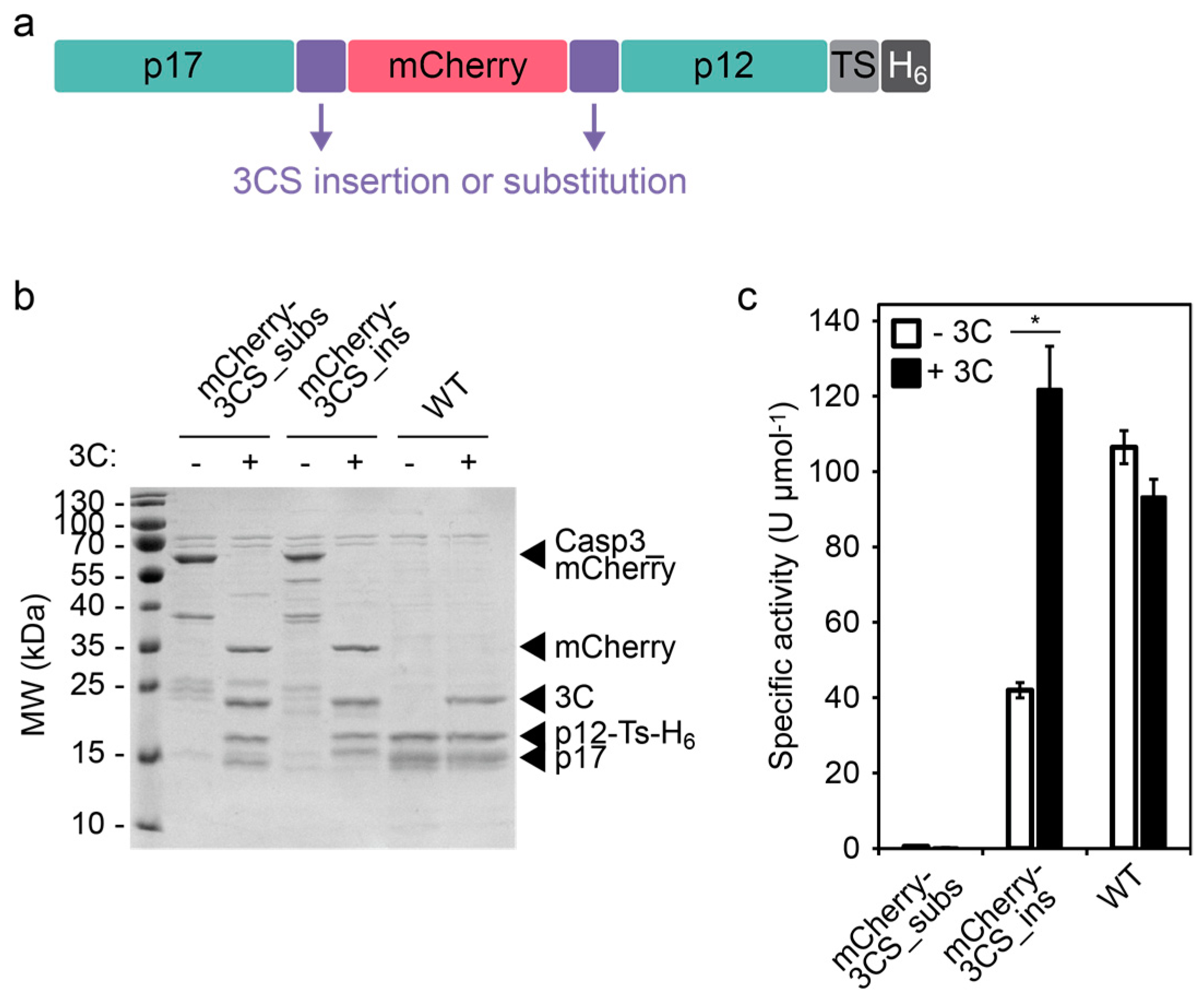
A new set of highly efficient, tag-cleaving proteases for purifying recombinant proteins - ScienceDirect

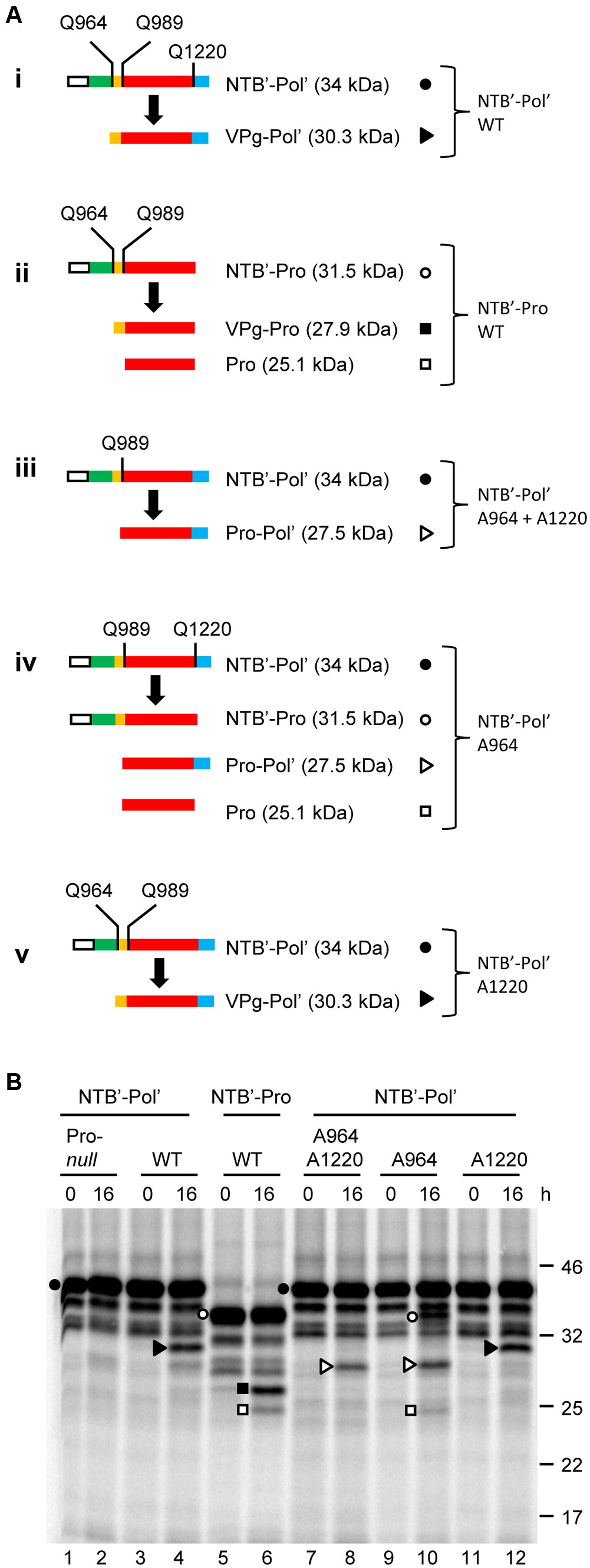
NT*-HRV3CP: An optimized construct of human rhinovirus 14 3C protease for high-yield expression and fast affinity-tag cleavage - ScienceDirect
Frontiers | Identification of Cleavage Sites Recognized by the 3C-Like Cysteine Protease within the Two Polyproteins of Strawberry Mottle Virus
Rhinovirus 3C Protease Facilitates Specific Nucleoporin Cleavage and Mislocalisation of Nuclear Proteins in Infected Host Cells | PLOS ONE

Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology
![Crystal structure of the 3C protease from Southern African Territories type 2 foot-and-mouth disease virus [PeerJ] Crystal structure of the 3C protease from Southern African Territories type 2 foot-and-mouth disease virus [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2016/1964/1/fig-1-full.png)
Crystal structure of the 3C protease from Southern African Territories type 2 foot-and-mouth disease virus [PeerJ]